
It provides a specific measure of the number of atoms or molecules in a bulk sample of matter. The mole is an amount unit similar to familiar units like pair, dozen, gross, etc. The three magnesium isotopes, Mg-24, Mg-25, and Mg-26, have atomic masses and relative abundance of. How can you find the average atomic mass of an element, if youre given the isotopic abundances and the mass of each isotope In nature, there are three types of magnesium: Mg-24 has a mass of 23.985 amu and accounts for 78.70 of all magnesium Mg-25 has a mass of 24.985 amu and accounts for 10. This experimental approach required the introduction of a new unit for amount of substances, the mole, which remains indispensable in modern chemical science. Solution for Calculate the atomic mass of magnesium. 
Today, we possess sophisticated instruments that allow the direct measurement of these defining microscopic traits however, the same traits were originally derived from the measurement of macroscopic properties (the masses and volumes of bulk quantities of matter) using relatively simple tools (balances and volumetric glassware). 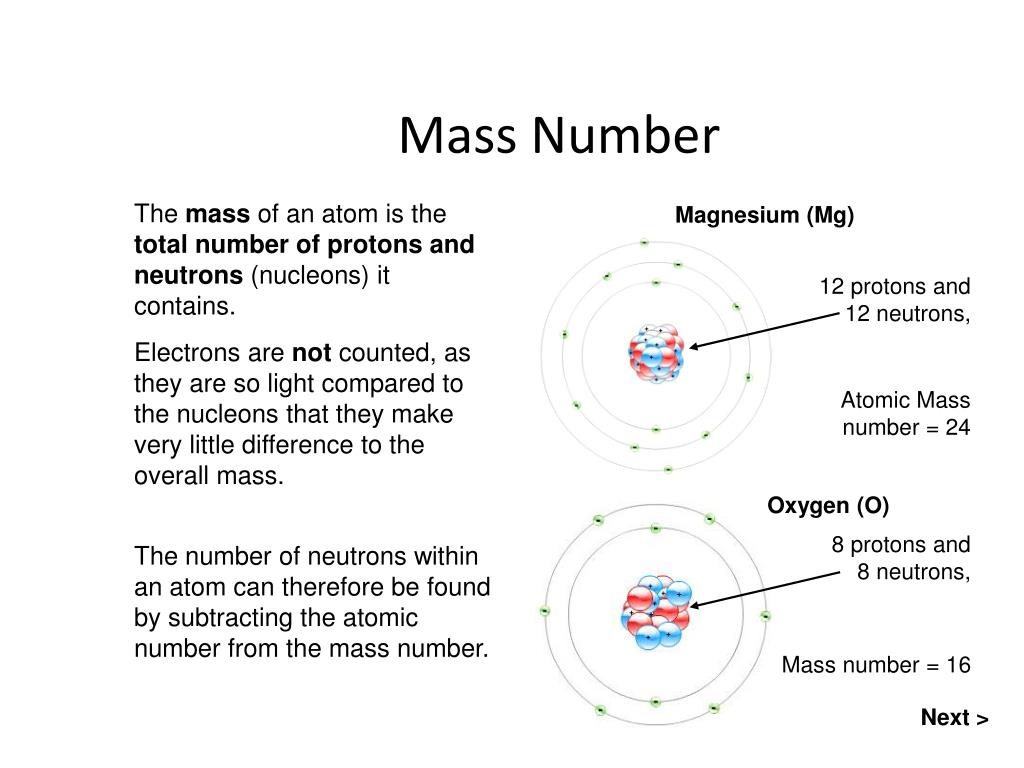
However, because a hydrogen peroxide molecule contains two oxygen atoms, as opposed to the water molecule, which has only one, the two substances exhibit very different properties. For example, water, H 2O, and hydrogen peroxide, H 2O 2, are alike in that their respective molecules are composed of hydrogen and oxygen atoms. Magnesium (Mg) Atomic Data for Magnesium (Mg) Atomic Number 12 Atomic Weight 24.3050 Reference E95 : Isotope : Mass : Abundance : Spin : Mag Moment : 24 Mg: 23.985042: 78.99: 0 : 25 Mg: 24.985837: 10.00: 5/2-0.85545: 26 Mg: 25.982593: 11.01: 0 : Mg I Ground State 1s 2 2s 2 2p 6 3s 2 1 S 0 Ionization energy 61671.05 cm-1 (7.646235 eV) Ref. The identity of a substance is defined not only by the types of atoms or ions it contains, but by the quantity of each type of atom or ion. Since its initial use during the development of modern atomic theory, MS has evolved to become a powerful tool for chemical analysis in a wide range of applications. The height of each vertical feature or peak in a mass spectrum is proportional to the fraction of cations with the specified mass-to-charge ratio. The ions are detected, and a plot of the relative number of ions generated versus their mass-to-charge ratios (a mass spectrum) is made. These cations then pass through a (variable) electric or magnetic field that deflects each cation’s path to an extent that depends on both its mass and charge (similar to how the path of a large steel ball bearing rolling past a magnet is deflected to a lesser extent that that of a small steel BB). \( \newcommand\)), the sample is vaporized and exposed to a high-energy electron beam that causes the sample’s atoms (or molecules) to become electrically charged, typically by losing one or more electrons.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
